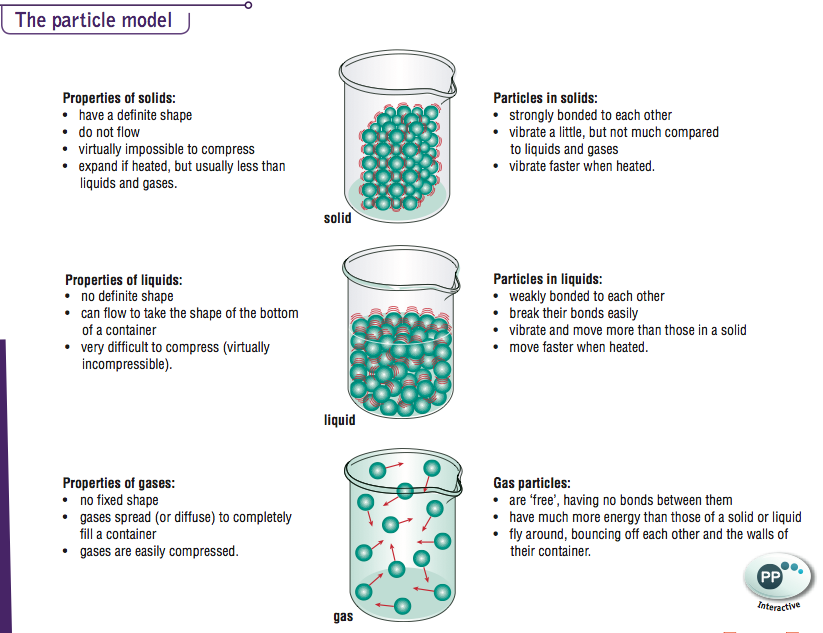
The molecules composing the gas are negligibly small compared to the distances between them.Gases are composed of molecules that are in continuous motion, travelling in straight lines and changing direction only when they collide with other molecules or with the walls of a container.(Note: The term “molecule” will be used to refer to the individual chemical species that compose the gas, although some gases are composed of atomic species, for example, the noble gases.) This theory is based on the following five postulates described here. The kinetic molecular theory (KMT) is a simple microscopic model that effectively explains the gas laws described in previous modules of this chapter. Although the gas laws describe relationships that have been verified by many experiments, they do not tell us why gases follow these relationships. The mathematical forms of these laws closely describe the macroscopic behavior of most gases at pressures less than about 1 or 2 atm.  
The gas laws that we have seen to this point, as well as the ideal gas equation, are empirical, that is, they have been derived from experimental observations. Use this theory’s postulates to explain the gas laws.State the postulates of the kinetic-molecular theory.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
